Artemether-Lumefantrine : Designed For Newborns
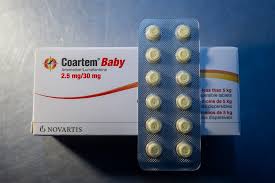
The World Health Organization (WHO) has cleared the first child-friendly version of artemether-lumefantrine specifically designed for newborns and infants weighing between 2 and 5 kg.
- A child-friendly formulation of a standard malaria therapy, now prequalified by the WHO for use in newborns and very young infants. Artemether-lumefantrine is a specialized two-drug combination therapy used to treat malaria by targeting the parasites in the bloodstream at different stages of their lifecycle.
- Aim is to close a long-standing treatment gap for babies weighing 2 to 5 kg, ensuring they receive quality-assured medicine instead of adjusted doses of adult or older-child formulations.
Key Features:
- Dual-Action Therapy: The medication combines two components: artemether, which acts rapidly to reduce the number of parasites in the blood, and lumefantrine, which stays in the system longer to clear remaining parasites and prevent the illness from returning.
- Precision Dosing: Unlike previous methods that required adjusting doses meant for older children—which risked harmful mistakes or side effects—this drug is specifically formulated for the weight class of newborns.
- This status allows global agencies and public health programs to procure the medicine reliably for high-burden regions.
- Specifically designed for the most vulnerable group—infants weighing as little as 2 kg.




