Einsteinium:
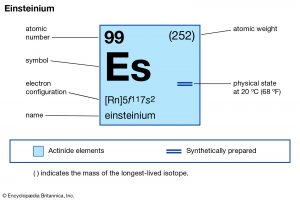
A team of scientists at the Berkeley Lab has reported some of the properties of element 99 in the periodic table called “Einsteinium”, named after Albert Einstein.
- With this new study published in the journal Nature last week, for the first time researchers have been able to characterize some of the properties of the element.
- It was discovered in 1952 in the debris of the first hydrogen bomb (the detonation of a thermonuclear device called “Ivy Mike” in the Pacific Ocean).
- The most common isotope of the element, einsteinium 253 has a half-life of 20 days.
- Because of its high radioactivity and short half-life of all einsteinium isotopes, even if the element was present on Earth during its formation, it has most certainly decayed.
- This is the reason that it cannot be found in nature and needs to be manufactured using very precise and intense processes.




